 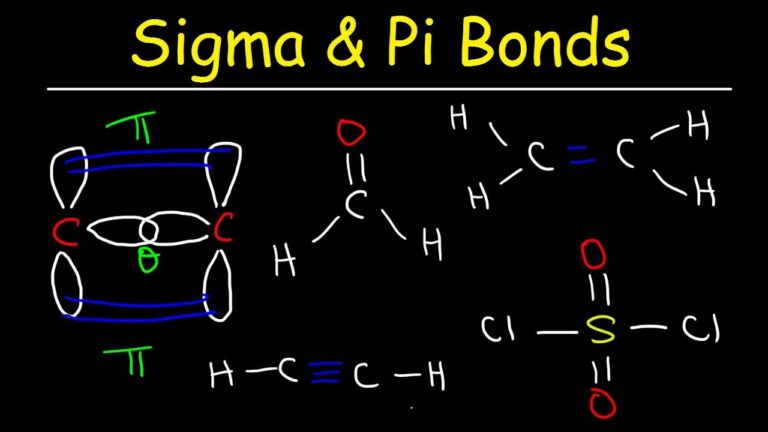
It is called inorganic benzene because it has 12 sigma bonds and 3 pi bonds the same as benzene. In ASP1H 2 O (A) and ASP2H 2 O (A) complexes the water molecule is interacting with the carbonyl oxygen O14 of the carboxylic acid group and the hydrogen bond distance is 1.98 and 2.11. Molecules whose Lewis structures can be drawn as a ring with alternating single and double bonds are known as annulenes.Annulenes are named by prefixing the number of carbon atoms in the rings in brackets before the word annulene: hence, benzene can be named as annulene. Quinoline consists of a benzene ring fused to the alpha-beta-position of the pyridine ring. This leads to 12 sigma bonds (do not forget the hydrogens) and 3 pi bonds. How do you determine the number of sigma and pi bonds? 4. There is also an addition bond called a pi bond present wherever there is a double bond. How many sigma bonds and how many pi-bonds are present in a benzene molecule? How many sigma and pi bonds does c6h6 have? Therefore 12- bonds and 3- bonds are present in Benzene molecule. How many sigma bonds and pi bonds are present in a benzene molecules ? Triple bonds are comprised of one sigma bond and two pi bonds. Einstein Mcfly said: Electrons are delocalized in benzene because a pi-bond NETWORK is created (if you want to draw the resonance structure of benzene, you'll see what I mean). (If the difference in electronegativity for the atoms in a bond is greater than 0.4, we consider the bond polar. The cyclopentadienyl anion is a planar, cyclic, regular-pentagonal ion it has 6 -electrons (4n 2, where n = 1), which fulfills Hckel's rule of aromaticity. How many $$\sigma$$ and $$\pi$$ bonds are present in the structure of D.D.T respectively? How many sigma and pi bonds are present in toluene? There are 20 sigma bonds present. Of sigma bond is the total count of C-C and C-H bonds phenanthrene has 10 C-H bonds and 16 C-C bondsso we have total 26 sigma bonds It have 14 carbon atoms and it`s a conjugated system so we`ll have 7 pi bonds here. Each carbon has 4 and each hydrogen 1 for a total of 10 electrons.The double bonds within this structure are mainly separated by a single bond, hence this arrangement is recognized to have conjugated double bonds. Add the valence electrons to the molecular orbital diagram.The 2p y orbitals on each carbon combine to make another 2 pi symmetry orbitals, 90 degrees from the first set. The 2p x orbitals on each atom combine to make 2 pi symmetry orbitals.(C-H bonds)Ĭombine the other 2 C(2sp) orbitals to make a sigma bonding and a sigma antibonding molecular orbital. Combine each H(1s) orbital with a C(2sp) orbital to make a sigma bonding and a sigma antibonding molecular orbital.After hybridization, a 2p x and a 2p y orbital remain on each carbon atom. The carbon atoms in ethyne use 2sp hybrid orbitals to make their sigma bonds.Each carbon atom makes 2 sigma bonds and has no lone pairs of electrons. Each carbon has 4 and each hydrogen 1 for a total of 12 electrons.Įthyne, sp hybridization with two pi bonds Finally, add the valence electrons to the molecular orbital diagram.The stabilization and destabilization in forming a pi bond are much less than for a sigma bond. The stabilization (decrease in energy) in going from the p orbital to pi bonding orbital equals the destabilization (increase in energy) in going from the p orbital to the pi antibonding orbital. These can combine to make a pi bonding and a pi antibonding molecular orbital. There remains a 2p orbital on each carbon.There are no remaining hybrid orbitals.(C-H bonds)Ĭombine the 2 C(2sp 2) orbitals to make a sigma bonding and a sigma antibonding molecular orbital. Combine each H(1s) orbital with a C(2sp 2) orbital to make a sigma bonding and a sigma antibonding molecular orbital.As with borane, make 2sp 2 hybrid orbitals on each carbon from the 2s, 2p x, and 2p y atomic orbitals.Each carbon forms 3 sigma bonds and has no lone pairs. The Lewis structure of the molecule CH 2CH 2 is below.They are not formed from hybrid orbitals. Pi bonds are formed from the overlap of parallel p orbitals on adjacent atoms. If a bond between two atoms is broken when one atom is rotated around the bond axis, that bond is called a pi bond. If two atoms are connected by a sigma bond, rotating one of the atoms around the bond axis doesn't break the bond. Sigma bonds are formed by the overlap of orbitals that are pointing directly towards one another. Hybrid orbitals are constructed from valence atomic orbitals and used to make sigma bonds between atoms. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
